Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 23.06.2019 00:00
What is the pressure of 0.500 moles of carbon dioxide gas in a 2.5 l tank and at a temperature of 301 k? (r=0.0821 l·atm/mol·k) 3.08 atm 1.2 atm 0.23 atm 4.01 atm 4.94 atm
Answers: 1

You know the right answer?
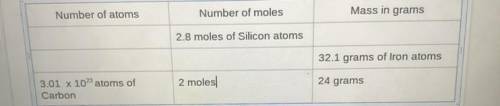
Can someone fill out the chart please?
...
...
Questions

English, 29.07.2019 18:00

Arts, 29.07.2019 18:00

Mathematics, 29.07.2019 18:00

Geography, 29.07.2019 18:00



Social Studies, 29.07.2019 18:00

English, 29.07.2019 18:00

English, 29.07.2019 18:00






Mathematics, 29.07.2019 18:00

Mathematics, 29.07.2019 18:00


History, 29.07.2019 18:00


Mathematics, 29.07.2019 18:00