
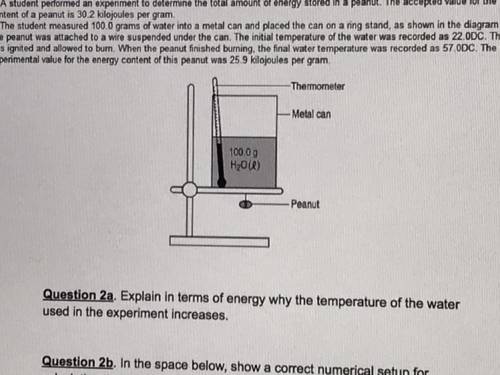
A student performed an experiment to determine the total amount of energy stored in a peanut. The accepted value for the energy content of a peanut is 30.2 kilojoules per gram . The student measured 100.0 grams of water into a metal can and placed the can on a ring stand, as shown in the diagram below. The peanut was attached to a wire suspended under the canThe initial temperature of the water was recorded as 22.0DC. The peanut was ignited and allowed to burn. When the peanut finished burning, the final water temperature was recorded as 57 ODC. The student's experimental value for the energy content of this peanut was 25.9 kilojoules per gram. Why the temperature of the water used in the experiment increase?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 23.06.2019 02:00
Pinene is an unsaturated hydrocarbon found in pine resin. if pinene has m+ = 136 and contains 1 double bond(s) and 2 ring(s); what is its molecular formula? enter the formula in the form ch first, then all other atoms in alphabetical order; do not use subscripts. the formula is case-sensitive
Answers: 3

Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2
You know the right answer?
A student performed an experiment to determine the total amount of energy stored in a peanut. The ac...
Questions

Social Studies, 23.08.2019 18:00

History, 23.08.2019 18:00

History, 23.08.2019 18:00



History, 23.08.2019 18:00

English, 23.08.2019 18:00





Mathematics, 23.08.2019 18:00


History, 23.08.2019 18:00




History, 23.08.2019 18:00




