
Chemistry, 02.12.2020 17:00 scottcourtney460
A biochemist carefully measures the molarity of magnesium ion in of cell growth medium to be . Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume of the cell growth medium falls to . Calculate the new molarity of magnesium ion in the cell growth medium. Be sure your answer has the correct number of significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:10
When the volume and number of particles of a gas are constant which of the following is also constant
Answers: 3

Chemistry, 22.06.2019 08:30
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 10:30
Aglow stick contains a glass vial with chemicals. when the glow stick is bent, the vial breaks and the chemicals react to produce a glow. a science student observes that a glow stick kept in the freezer glows for a longer duration than a glow stick kept at room temperature. what conclusion can be drawn based on the observation? be sure to note the outcome and test variables in the conclusion.
Answers: 1

You know the right answer?
A biochemist carefully measures the molarity of magnesium ion in of cell growth medium to be . Unfor...
Questions





Mathematics, 28.05.2020 05:00

Mathematics, 28.05.2020 05:00

Mathematics, 28.05.2020 05:00







Mathematics, 28.05.2020 05:00




Mathematics, 28.05.2020 05:00

Chemistry, 28.05.2020 05:00

Mathematics, 28.05.2020 05:00

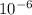 x 47 x
x 47 x 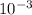 = 4.559 x
= 4.559 x 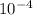 M = 759.83333 uM
M = 759.83333 uM

