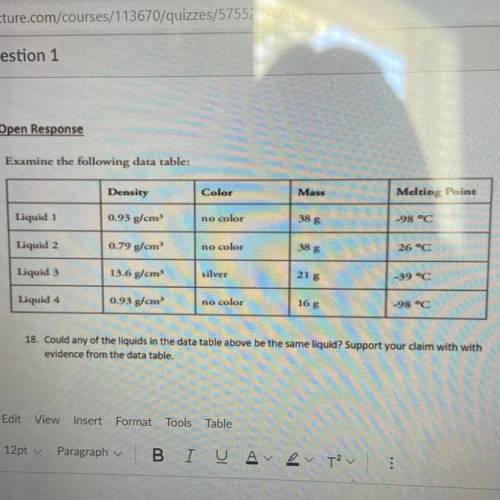
Could someone pls help me :)
...

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 02:30
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2


Chemistry, 23.06.2019 09:30
Which of the following is not a characteristic of a hydrogen bond? 1. it is responsible for the unusual physical properties of water. 2. it is weaker than a covalent bond. 3. it is stronger than other dipole-dipole interactions. 4. it can occur when hydrogen is covalently bound to very electronegative elements liks f, cl, br and i.
Answers: 1

Chemistry, 23.06.2019 17:20
What is the relationship between ka and kb with kw? the sum of ka and kb equals the auto-dissociation constant for water. the product of ka and kb equals the auto-dissociation constant for water. the quotient of ka and kb equals the auto-dissociation constant for water. the difference of ka and kb equals the auto-dissociation constant for water.
Answers: 3
You know the right answer?
Questions

History, 23.08.2019 05:30


Mathematics, 23.08.2019 05:30


Health, 23.08.2019 05:30


Mathematics, 23.08.2019 05:30

Chemistry, 23.08.2019 05:30

Social Studies, 23.08.2019 05:30





Geography, 23.08.2019 05:30





Computers and Technology, 23.08.2019 05:30