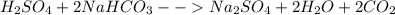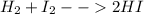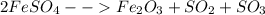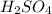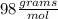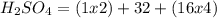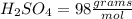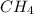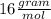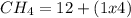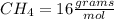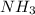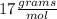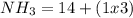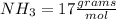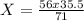Balance the following equations:
i. H2SO4 + ….NaHCO3 → Na2SO4 + ….H2O + ……CO2
ii. H2 + I2 → ….HI
iii. …..NaOH + H2SO4 → Na2SO4 + …..H2O
iv. ……FeSO4 → Fe2O3 + SO2 + SO3
v. C3H6O2 + …..O2 → ….CO2 + …..H2O
b) Calculate the molecular masses of the following:
i. H2SO4
ii. CH4
iii. NH3
c) 56g of iron reacts with 71g of chlorine. How many grams of iron react with 35.5g of chlorine?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 3

Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 12:30
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1
You know the right answer?
Balance the following equations:
i. H2SO4 + ….NaHCO3 → Na2SO4 + ….H2O + ……CO2
ii. H2 + I2 → …....
ii. H2 + I2 → …....
Questions


Mathematics, 01.06.2020 03:58



Business, 01.06.2020 03:58

Chemistry, 01.06.2020 03:58

Mathematics, 01.06.2020 03:58

English, 01.06.2020 03:58

Social Studies, 01.06.2020 03:58


Chemistry, 01.06.2020 03:58


Mathematics, 01.06.2020 03:58


Mathematics, 01.06.2020 03:58

Mathematics, 01.06.2020 03:58

Mathematics, 01.06.2020 03:58

History, 01.06.2020 03:58

Mathematics, 01.06.2020 03:58

Law, 01.06.2020 03:58