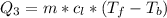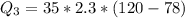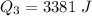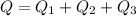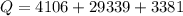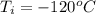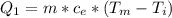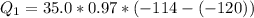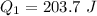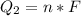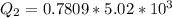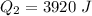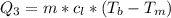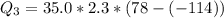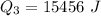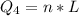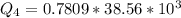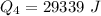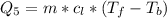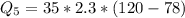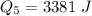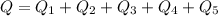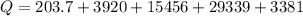Ethanol (C2H5OH) melts at –114 °C and boils at 78 °C. The enthalpy of fusion of ethanol is 5.02 kJ/mol, and its enthalpy of vaporization is 38.56 kJ/mol. The specific heats of solid and liquid ethanol are 0.97 J/g-K and 2.3 J/g-K, respectively. The average specific heat of gaseous ethanol is about 1.80 J/g-K. a. How much heat is required to convert 35.0 g of ethanol at 27 °C to the vapor phase at 120 °C? b. How much heat is required to convert the same amount of ethanol at –120 °C to the vapor phase at 120 °C?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 14:00
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3

You know the right answer?
Ethanol (C2H5OH) melts at –114 °C and boils at 78 °C. The enthalpy of fusion of ethanol is 5.02 kJ/m...
Questions



Mathematics, 07.12.2020 23:20



Computers and Technology, 07.12.2020 23:20


Arts, 07.12.2020 23:20


Health, 07.12.2020 23:20




Arts, 07.12.2020 23:20



Mathematics, 07.12.2020 23:20



Mathematics, 07.12.2020 23:20

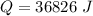
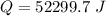
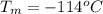
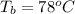
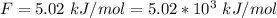
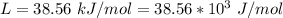
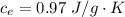
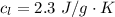
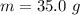
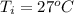
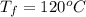
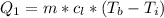
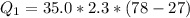
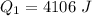
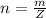
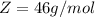
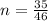
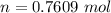
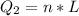
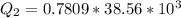
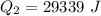
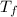 is mathematically represented as
is mathematically represented as