
Chemistry, 04.12.2020 02:20 davidleew24
An atom of element Be has four protons, four electrons, and five neutrons. Which element below could be an isotope of this atom?
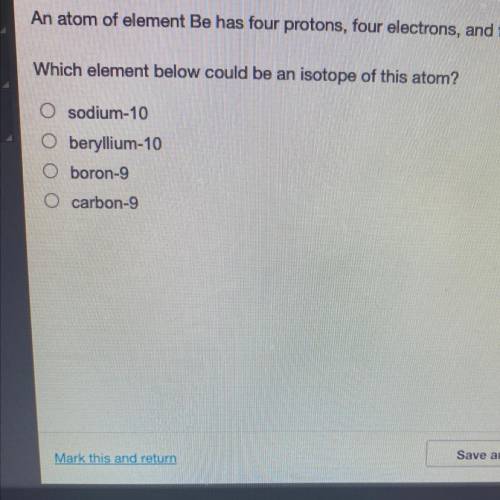

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1


Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 13:30
The atomic number, or number, is the described as the number of in the nucleus of an chemical element.
Answers: 1
You know the right answer?
An atom of element Be has four protons, four electrons, and five neutrons.
Which element below coul...
Questions

Spanish, 23.02.2021 17:40



Mathematics, 23.02.2021 17:40



Chemistry, 23.02.2021 17:40


Mathematics, 23.02.2021 17:40

Mathematics, 23.02.2021 17:40

Mathematics, 23.02.2021 17:40



Biology, 23.02.2021 17:40

Mathematics, 23.02.2021 17:40


Mathematics, 23.02.2021 17:40


Mathematics, 23.02.2021 17:40

Biology, 23.02.2021 17:40



