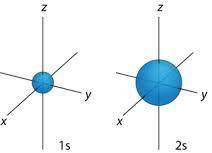
The pair "2s and 1s" has the same shape. A complete explanation is below.
Throughout Mendeleev's table, the "s" orbital has a sphere as well as round symmetry. This same wave function would be determined by the difference again from the nucleus rather than the directions.These same 2s, as well as 1s orbitals, are approximately identical in form, with the exception being the 2s orbital appears bigger than the whole 1s orbital.
Thus the above alternative i.e., "option B" is appropriate.
Learn more:
link