Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
What are the variables in gay-lussac’s law? pressure and volume pressure, temperature, and volume pressure and temperature volume, temperature, and moles of gas
Answers: 1

Chemistry, 23.06.2019 18:20
Ahydrogen electron is elevated from level 1 to level 2. another electron is elevated from level 2 to level 4. the transition requiring the greatest energy change is?
Answers: 1


Chemistry, 23.06.2019 22:00
Which sentence best explains the high melting point, boiling point, and surface tension of water? a. the hydrogen atom on each water molecule is strongly attracted to the hydrogen atoms on nearby molecules. b. ions dissolved in the water cause nearby molecules to become temporarily polar. c. the water molecules are connected to each other with covalent bonds. d. water molecules that have lost electrons attract the water molecules that have gained electrons. e. the negative side of each water molecule is attracted to the positive sides of nearby molecules.
Answers: 1
You know the right answer?
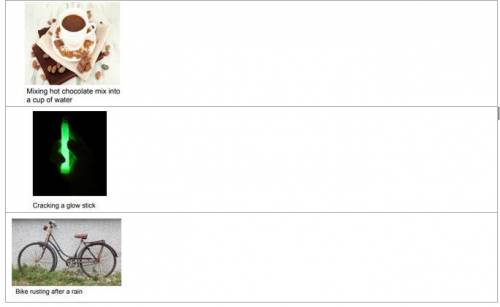
Is this a chemical reaction or not? Explain why you think so (Be sure to answer both parts of the qu...
Questions

Mathematics, 11.11.2020 23:30


Mathematics, 11.11.2020 23:30


Mathematics, 11.11.2020 23:30

Mathematics, 11.11.2020 23:30


Spanish, 11.11.2020 23:30




History, 11.11.2020 23:30

English, 11.11.2020 23:30

English, 11.11.2020 23:30

Mathematics, 11.11.2020 23:30


Physics, 11.11.2020 23:30


Mathematics, 11.11.2020 23:30

Mathematics, 11.11.2020 23:30