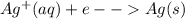Chemistry, 28.08.2019 21:40 alysiareyes
If you needed to make ultrapure silver from crude silver, where would you collect the pure silver metal in an electrolytic cell?
a. cathode
b. anode
c. connecting wires
d. power source

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 23:10
Match the formula for the following compound: magnesium sulfate heptahydratemgs·7h2omg2so4·7h2omg(so4)2·7h2omgso4·7h2o
Answers: 1

Chemistry, 23.06.2019 01:20
How can parts of a solution be separated by chromatography?
Answers: 1
You know the right answer?
If you needed to make ultrapure silver from crude silver, where would you collect the pure silver me...
Questions

Biology, 30.11.2019 20:31

Mathematics, 30.11.2019 20:31

History, 30.11.2019 20:31

Physics, 30.11.2019 20:31


Mathematics, 30.11.2019 20:31

English, 30.11.2019 20:31

Health, 30.11.2019 20:31



Chemistry, 30.11.2019 20:31

Mathematics, 30.11.2019 20:31


Chemistry, 30.11.2019 20:31

Biology, 30.11.2019 20:31

Advanced Placement (AP), 30.11.2019 20:31


Mathematics, 30.11.2019 20:31

History, 30.11.2019 20:31

Chemistry, 30.11.2019 20:31