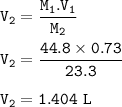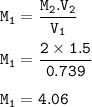Chemistry, 05.12.2020 14:00 najeezubair0666
Part 1: What is the final volume in milliliters when 0.730 L of a 44.8 % (m/v) solution is diluted to 23.3 % (m/v)? part 2:A 739 mL NaCl solution is diluted to a volume of 1.50 L and a concentration of 2.00 M . What was the initial concentration?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 09:30
Melissa is interested in her family tree and how her family has changed over its many generations. melissa probably more closely resembles
Answers: 2

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
You know the right answer?
Part 1: What is the final volume in milliliters when 0.730 L of a 44.8 % (m/v) solution is diluted t...
Questions








Mathematics, 23.04.2021 21:00

Mathematics, 23.04.2021 21:00

Mathematics, 23.04.2021 21:00

Mathematics, 23.04.2021 21:00


Mathematics, 23.04.2021 21:00


Mathematics, 23.04.2021 21:00

Mathematics, 23.04.2021 21:00

History, 23.04.2021 21:00


Mathematics, 23.04.2021 21:00