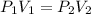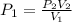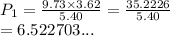A sample of argon, Ar, has a volume of 5.40 L with an unknown pressure. The gas has a volume of 9.73 L when the pressure is 3.62 atm,
with no change in temperature and amount of gas. What was the initial pressue in atm of the gas?
O a. 6.523 atm
O b. 6.52 atm
Oc 2.009 atm
Od. 2,01 atm

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 16:50
Ajet plane is speeding down the runway during takeoff. air resistance is not negligible. identify the forces on the jet.
Answers: 3

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
A sample of argon, Ar, has a volume of 5.40 L with an unknown pressure. The gas has a volume of 9.73...
Questions

Biology, 07.10.2019 19:30

Mathematics, 07.10.2019 19:30



Health, 07.10.2019 19:30



Mathematics, 07.10.2019 19:30

Spanish, 07.10.2019 19:30

Biology, 07.10.2019 19:30

Mathematics, 07.10.2019 19:30

English, 07.10.2019 19:30


Mathematics, 07.10.2019 19:30

Chemistry, 07.10.2019 19:30


Mathematics, 07.10.2019 19:30