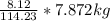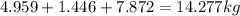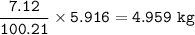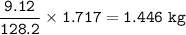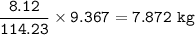Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Si una estrella no tiene paralaje medible, ¿qué puedes inferir?
Answers: 1


Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1

Chemistry, 22.06.2019 14:00
How is the atomic number of a nucleus changed by alpha decay
Answers: 2
You know the right answer?
A mixture of hydrocarbons contains 34.8% heptane, C7H16, 10.1% nonane, C9H20, and 55.1% octane, C8H1...
Questions


Chemistry, 09.06.2020 18:57


Mathematics, 09.06.2020 18:57



Mathematics, 09.06.2020 18:57

Mathematics, 09.06.2020 18:57

Mathematics, 09.06.2020 18:57





Mathematics, 09.06.2020 18:57

Mathematics, 09.06.2020 18:57





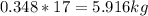 Mass C in Heptane (MW=100.21 g/mol) :
Mass C in Heptane (MW=100.21 g/mol) :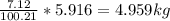
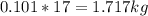 Mass C in Nonane (MW=128.2 g/mol) :
Mass C in Nonane (MW=128.2 g/mol) :
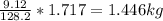
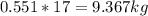 Mass C in Otane (MW=114.23 g/mol) :
Mass C in Otane (MW=114.23 g/mol) :