
Chemistry, 06.12.2020 22:10 yasminothman02
Calculate the atomic mass of each of the following elements using your method from above. Test your answer using the Nature’s mix of isotopes and the periodic table. Keep going until you can get two in a row right.
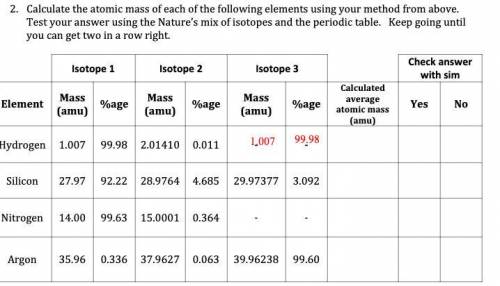

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2


Chemistry, 23.06.2019 02:30
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1

Chemistry, 23.06.2019 10:30
4al + 3o2 → 2al2o3 what does the "3" in front of o2 stand for? a) it indicates that there are 5 oxygen atoms after you add the coefficient and the subscript. b) it indicates that that there are are total of 6 oxygen atoms all bonded together as a single molecule. c) it indicates that there are 3 oxygen molecules chemically bonded to each other in the reaction. d) it indicates that there are 3 separate oxygen molecules in the reaction.
Answers: 2
You know the right answer?
Calculate the atomic mass of each of the following elements using your method from above. Test your...
Questions

Social Studies, 29.10.2020 01:00



English, 29.10.2020 01:00



History, 29.10.2020 01:00



Mathematics, 29.10.2020 01:00




History, 29.10.2020 01:00

Spanish, 29.10.2020 01:00

Mathematics, 29.10.2020 01:00

Health, 29.10.2020 01:00


Mathematics, 29.10.2020 01:00

SAT, 29.10.2020 01:00




