
Chemistry, 08.12.2020 05:20 alexusnicole817
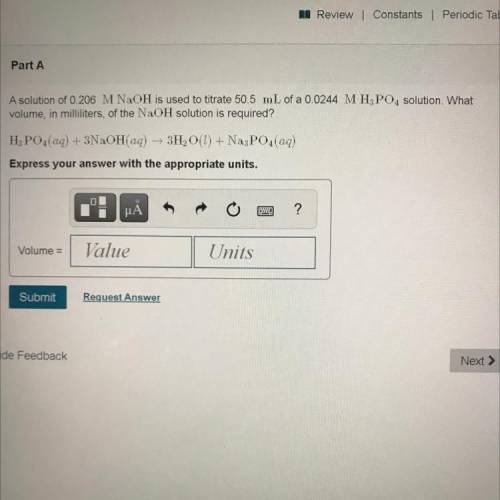
Part A
A solution of 0.206 M NaOH is used to titrate 50.5 mL of a 0.0244 MH3PO4 solution. What
volume, in milliliters, of the NaOH solution is required?
H3PO4(aq) + 3NaOH(aq) + 3H2O(l) + Na3PO4(aq)
Express your answer with the appropriate units.
μΑ
?
Volume =
Value
Units
Submit
Request Answer
Provide eedback

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:00
What is the molality of a solution that has 4 mol of kci in 0.800 kg of water
Answers: 3

Chemistry, 21.06.2019 20:30
In a laboratory experiment, a fermenting aqueous solution of glucose and yeast produces carbon dioxide gas and ethanol. the solution was heated by burning natural gas in a bunsen burner to distill the ethanol that formed in the flask. during the distillation, the ethanol evaporated and then condensed in the receiving flask. the flame of the burner was kept too close to the bottom of the flask and some of the glucose decomposed into a black carbon deposit on the inside of the flask. during this experiment the following changes occurred. which of these changes involved a physical change and not a chemical change? check all that apply. 1-condensation of ethanol 2-evaporation of ethanol 3- formation of carbon dioxide gas from glucose burning of natural gas 4-formation of ethanol from glucose by yeast 5-formation of a carbon deposit inside the flask
Answers: 2


Chemistry, 22.06.2019 01:00
Water is important for the of cells. a: size, shape, and temperature b: temperature, color, and odor c: color, odor, and size d: shape, temperature, and color
Answers: 2
You know the right answer?
Part A
A solution of 0.206 M NaOH is used to titrate 50.5 mL of a 0.0244 MH3PO4 solution. What
Questions


English, 07.09.2021 03:00



Mathematics, 07.09.2021 03:00


Chemistry, 07.09.2021 03:00


Mathematics, 07.09.2021 03:00


Chemistry, 07.09.2021 03:00




English, 07.09.2021 03:10




English, 07.09.2021 03:10

Biology, 07.09.2021 03:10



