Sheet may help you to answer
his questio
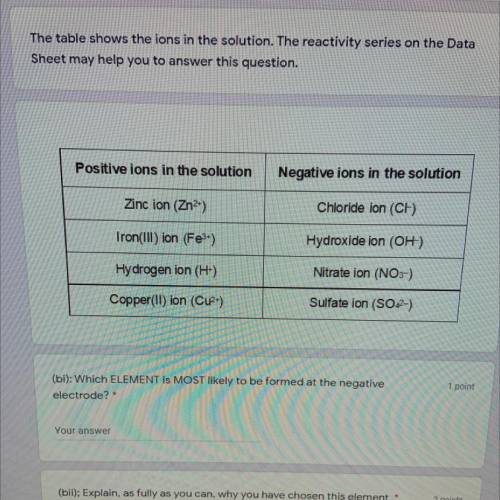
Positive ions
the solution
Negative ions...

Sheet may help you to answer
his questio
Positive ions
the solution
Negative ions in the solution
Zinc ion (Zn2+)
Chloride ion (CH)
Iron(Ill) ion (Fe3+)
Hydroxide ion (OH)
Hydrogen ion (H+)
Nitrate ion (NO3-)
Copper(II) ion (Cuz)
Sulfate ion (SO42-)
(bi): Which ELEMENT IS MOST likely to be formed at the negative
electrode?
1 point
Your answer

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 09:00
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 17:10
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1
You know the right answer?
Questions





Mathematics, 25.06.2019 13:30










Mathematics, 25.06.2019 13:30


Mathematics, 25.06.2019 13:30

History, 25.06.2019 13:30




