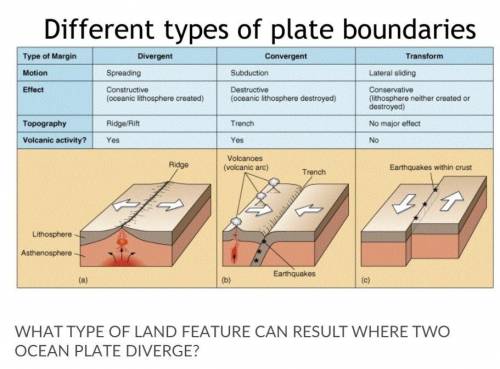
A mountain
b trench
c mid ocean range
...

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 23:30
Match each statement with the state of matter it describes
Answers: 3

Chemistry, 23.06.2019 01:40
Calcium carbonate decomposes at high temperatures to give calcium oxide and carbon dioxide as shown below. caco3(s) cao(s) + co2(g) the kp for this reaction is 1.16 at 800°c. a 5.00 l vessel containing 10.0 g of caco3(s) was evacuated to remove the air, sealed, and then heated to 800°c. ignoring the volume occupied by the solid, what will be the mass of the solid in the vessel once equilibrium is reached?
Answers: 1

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1

Chemistry, 23.06.2019 05:00
110 g of water (specific heat = 4.184 j/g c) and 100 g of a metal sample (specific heat = 0.397 j/g c) are heated from 25 degrees c to 75 degrees c. which substance required more thermal energy?
Answers: 1
You know the right answer?
Questions

Health, 18.03.2021 02:30

English, 18.03.2021 02:30



Computers and Technology, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

History, 18.03.2021 02:30

English, 18.03.2021 02:30


Mathematics, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

Advanced Placement (AP), 18.03.2021 02:30



Geography, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30

Mathematics, 18.03.2021 02:30