
Chemistry, 09.12.2020 20:00 jakobrobinette
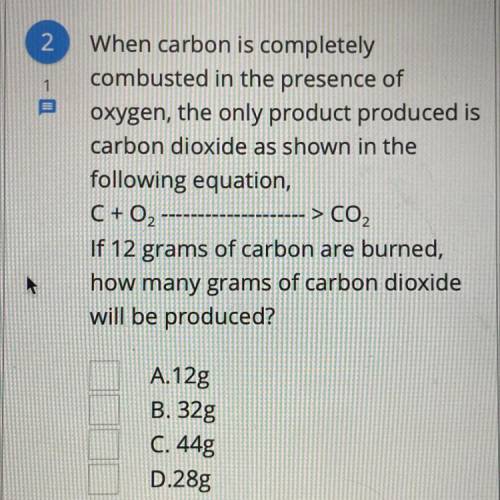
When carbon is completely combusted in the presence of oxygen the only product produced is carbon dioxide as shown in the following equation, C+O2——>CO2 If 12 grams of carbon are burned, how many grams of carbon dioxide will be produced?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
The isotonic saline solution described in part a is connected to an unknown solution via a semipermeable membrane, the unknown solution level drops. based on this information, what can be said about these two solutions?
Answers: 1

Chemistry, 22.06.2019 01:30
Idon't really understand this can you me and show your work.☺☺[ chemistry b] subject [ electron transfer in lonic bonds]grade( 12)
Answers: 1

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
You know the right answer?
When carbon is completely combusted in the presence of oxygen the only product produced is carbon di...
Questions

Mathematics, 04.03.2021 01:00




Physics, 04.03.2021 01:00


Mathematics, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00

English, 04.03.2021 01:00


Biology, 04.03.2021 01:00

Mathematics, 04.03.2021 01:00

English, 04.03.2021 01:00

Biology, 04.03.2021 01:00



Mathematics, 04.03.2021 01:00

History, 04.03.2021 01:00




