10 Balance the following equation: + CO2 (g) + H2O (1) → C12H24012 (s) + O2 (g)
...

Chemistry, 10.12.2020 03:00 starlodgb1971
10 Balance the following equation: + CO2 (g) + H2O (1) → C12H24012 (s) + O2 (g)
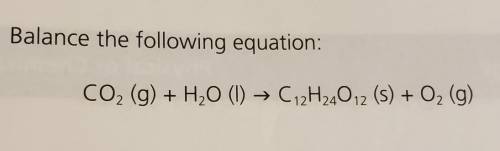

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:40
How many liters of hydrogen gas will be produced at stp from the reaction of 7.179×10^23 atoms of magnesium with 54.219g of phosphoric acid (h3po4) the equation is 3mg + 2h3(> mg(po4)2+3h2
Answers: 1

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 09:30
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3
You know the right answer?
Questions



History, 22.11.2019 23:31


Mathematics, 22.11.2019 23:31



History, 22.11.2019 23:31


Mathematics, 22.11.2019 23:31

Mathematics, 22.11.2019 23:31

Business, 22.11.2019 23:31

Mathematics, 22.11.2019 23:31

Mathematics, 22.11.2019 23:31

Biology, 22.11.2019 23:31



Social Studies, 22.11.2019 23:31

Mathematics, 22.11.2019 23:31

Mathematics, 22.11.2019 23:31



