
Ammonia (A) diffuses through a stagnant layer of air (B), 1cm thick, at 25 ºC and 1 atm total pressure. The partial pressures of ammonia on the two sides of the air layer are: PA0=0.9 atm and PAl=0.1 atm respectively. Air is none diffusing. Calculate the molar flux of ammonia. DAB= 0.214 cm2 /s

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 06:00
An atom of sodium-23 (atomic number = 11) has a positive charge of +1. give this information, how many electrons does it have? how many proteins and neutrons does this atom have
Answers: 2

Chemistry, 23.06.2019 08:40
Calculate the number of grams of sodium in 3.00 g of each sodium-containing food additive.
Answers: 3

Chemistry, 23.06.2019 11:00
Which example is a mechanical wave? a.microwave b.radio wave c.water wave d.ultraviolet light
Answers: 1
You know the right answer?
Ammonia (A) diffuses through a stagnant layer of air (B), 1cm thick, at 25 ºC and 1 atm total pressu...
Questions



Biology, 26.05.2020 02:02

History, 26.05.2020 02:02

Mathematics, 26.05.2020 02:02









Mathematics, 26.05.2020 02:02





French, 26.05.2020 02:02

History, 26.05.2020 02:02

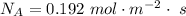
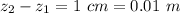
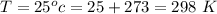
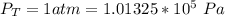
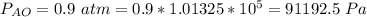
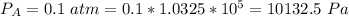
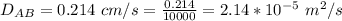
![N_A = \frac{D_{AB} * P_T }{RT(z_2 -z_1)} * ln [\frac{P_T - P_{Al}}{P_T - P_{AO}} ]](/tpl/images/0969/5009/086d8.png)
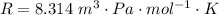
![N_A = \frac{2.14 *10^{-5} * 1.01325*10^{5} }{8.314 *298 (0.01)} * ln [\frac{1 - 0.1}{1 - 0.9} ]](/tpl/images/0969/5009/c7bba.png)


