
Chemistry, 12.12.2020 17:10 jakhunter354
Indicate whether aqueous solutions of each of the following solutes will contain only ions, only molecules, or mostly molecules and a few ions:
a) NH4Cl, a strong electrolyte.
b) Ethanol, CH3-CH2-OH, a nonelectrolyte.
c) HCN, hydrocyanic acid, a weak electrolyte.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2

Chemistry, 23.06.2019 00:00
What does an electron configuration for an atom relate to the atoms placement on the periodic table
Answers: 2

Chemistry, 23.06.2019 03:00
Air pressure is measured in pascals. for a professional american football game, the ball should be inflated to about 90,000 pascals. scientists studied the effects of air temperature on the pressure inside american footballs by taking these steps: 1. prepare 100 footballs. 2. measure each football's air pressure. 3. divide footballs into 10 groups. 4. place the groups in different lockers cooled to different air temperatures. 5. after 12 hours, remove the footballs from lockers. 6. measure each football's pressure again. 7. compare the new pressures to the starting pressures. what two terms best describe the variable "air pressure inside the football" in this experiment? independent, qualitative independent, quantitative dependent, qualitative dependent, quantitative
Answers: 3
You know the right answer?
Indicate whether aqueous solutions of each of the following solutes will contain only ions, only mol...
Questions



Biology, 30.11.2021 02:10


English, 30.11.2021 02:10





Mathematics, 30.11.2021 02:10

English, 30.11.2021 02:10

Computers and Technology, 30.11.2021 02:10

Business, 30.11.2021 02:10



SAT, 30.11.2021 02:10

Mathematics, 30.11.2021 02:10


Social Studies, 30.11.2021 02:10

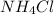 has a strong electrolyte.
has a strong electrolyte.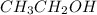 , it has a nonelectrolyte.
, it has a nonelectrolyte.

