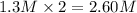Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3


Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1

Chemistry, 23.06.2019 02:00
Which of the following substances is the most soluble in water? a. sodium chloride b. methane c. bromine d. carbon
Answers: 1
You know the right answer?
What are the respective concentrations (M) of Mg2+and Cl-afforded by dissolving 0.325 mol MgCl2 in w...
Questions




Health, 13.11.2020 23:00


English, 13.11.2020 23:00

Mathematics, 13.11.2020 23:00


Biology, 13.11.2020 23:00




Spanish, 13.11.2020 23:00

Mathematics, 13.11.2020 23:00




Mathematics, 13.11.2020 23:00

Mathematics, 13.11.2020 23:00

English, 13.11.2020 23:00

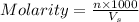
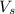 = volume of solution in ml
= volume of solution in ml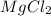 =
= 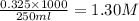
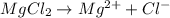
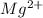 and two moles of
and two moles of 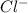 ions .
ions .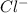 =
=