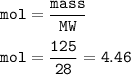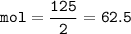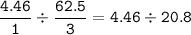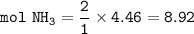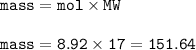Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 23.06.2019 03:30
If you need to add 27.50ml of a solution, which piece of glassware would you use to deliver this volume and explain how you would determine if the 27.50 ml was measured?
Answers: 1

Chemistry, 23.06.2019 04:00
What is the volume of 2.5 moles of nitrogen gas (n2)at standard temperature and pressure (stp)?
Answers: 1

Chemistry, 23.06.2019 05:50
What is the molecular formula of ferrous nitrate and ferric nitrate
Answers: 2
You know the right answer?
If 125.0g of nitrogen is reacted with 125.0g of hydrogen, what is the theoretical yield of the react...
Questions

Chemistry, 24.09.2019 12:30


Biology, 24.09.2019 12:30


Chemistry, 24.09.2019 12:30


Mathematics, 24.09.2019 12:30

Mathematics, 24.09.2019 12:30


Chemistry, 24.09.2019 12:30

Mathematics, 24.09.2019 12:30

Biology, 24.09.2019 12:30

Chemistry, 24.09.2019 12:30


Health, 24.09.2019 12:30

Arts, 24.09.2019 12:30

Mathematics, 24.09.2019 12:30


Social Studies, 24.09.2019 12:30