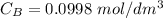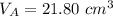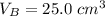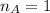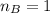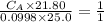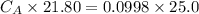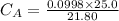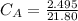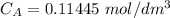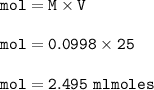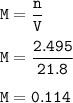Student carries out a titration to determine the concentration of a solution of
nitric acid. She titrates the solution of nitric acid against a standard solution
of sodium hydroxide with a known concentration of 0.0998 mol/dm². She
finds that 21.80 cm of the nitric acid solution is needed to exactly neutralise
25.0 cm of the sodium hydroxide solution.
Calculate the concentration of the nitric acid solution. Give your answer to
three significant figures.
The equation for the neutralisation reaction is
HNO3 + NaOH → NaNO3 + H2O

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:00
If a reaction has g of -136kj at 110°c, will it be spontaneous at this temperature (110°c)? yes or no
Answers: 2

Chemistry, 21.06.2019 21:30
An alcohol thermometer makes use of alcohol's changing in order to measure temperature. as the temperature goes up, the alcohol contained in the thermometer increases in volume, filling more of the thermometer's tube.
Answers: 3

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

You know the right answer?
Student carries out a titration to determine the concentration of a solution of
nitric acid. She ti...
Questions

Biology, 23.09.2019 07:30

Computers and Technology, 23.09.2019 07:30

Mathematics, 23.09.2019 07:30

History, 23.09.2019 07:30


Mathematics, 23.09.2019 07:30



Mathematics, 23.09.2019 07:30

Mathematics, 23.09.2019 07:30



Mathematics, 23.09.2019 07:30

Mathematics, 23.09.2019 07:30




Biology, 23.09.2019 07:30

Mathematics, 23.09.2019 07:30

Mathematics, 23.09.2019 07:30

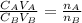
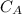 is the concentration of acid
is the concentration of acid
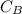 is the concentration of base
is the concentration of base
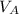 is the volume of acid
is the volume of acid
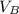 is the volume of base
is the volume of base
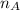 is the mole ratio of acid
is the mole ratio of acid
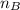 is the mole ratio of base
is the mole ratio of base