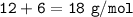Chemistry, 15.12.2020 14:20 arianasg06
Calculate the mass of atoms of carbon and 3 molecules of hydrogen

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 21.06.2019 18:30
Strong conductivity of plasma allows it to act and react as and
Answers: 2

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3
You know the right answer?
Calculate the mass of atoms of carbon and 3 molecules of hydrogen...
Questions




Mathematics, 26.11.2019 08:31


Biology, 26.11.2019 09:31

World Languages, 26.11.2019 09:31

Social Studies, 26.11.2019 09:31

Mathematics, 26.11.2019 09:31

Mathematics, 26.11.2019 09:31

Mathematics, 26.11.2019 09:31

English, 26.11.2019 09:31

Mathematics, 26.11.2019 09:31


Mathematics, 26.11.2019 09:31

Mathematics, 26.11.2019 09:31

Mathematics, 26.11.2019 09:31

English, 26.11.2019 09:31

History, 26.11.2019 09:31

Mathematics, 26.11.2019 09:31