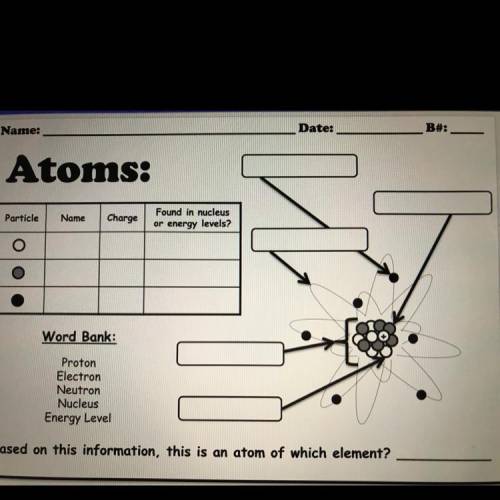
Please I need help I really don’t get this lol
...

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 17:10
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2


Chemistry, 22.06.2019 18:10
Measurements that have similar values are: a. usually accurate b. sometimes accurate c. always accurate d. never accurate
Answers: 1

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1
You know the right answer?
Questions




Mathematics, 16.07.2019 18:00



Mathematics, 16.07.2019 18:00


History, 16.07.2019 18:00

History, 16.07.2019 18:00





English, 16.07.2019 18:00

Biology, 16.07.2019 18:00


Mathematics, 16.07.2019 18:00

Chemistry, 16.07.2019 18:00

Mathematics, 16.07.2019 18:00