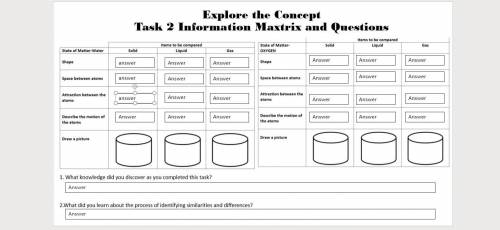
Plsplsplsplsplsplsplsplsplspls help
...

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 12:30
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 20:20
Which symbol can be used to indicate the pressure at which a chemical reaction is carried out? 25°c 2 atm pa
Answers: 2
You know the right answer?
Questions




History, 21.01.2021 01:00


English, 21.01.2021 01:00


Mathematics, 21.01.2021 01:00

Chemistry, 21.01.2021 01:00

Social Studies, 21.01.2021 01:00

English, 21.01.2021 01:00

Mathematics, 21.01.2021 01:00

History, 21.01.2021 01:00

Social Studies, 21.01.2021 01:00


Biology, 21.01.2021 01:00

Mathematics, 21.01.2021 01:00



Mathematics, 21.01.2021 01:00