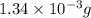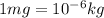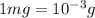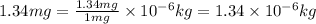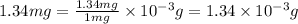Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
If you chip a tooth, most likely you will go to the dentist to have the missing material filled in. currently the material used to fill in teeth is a polymer that is flexible when put in, yet is hardened to the strength of a tooth after irradiation with blue light at a wavelength of 461 nm. what is the energy in joules for a photon of light at this wavelength?
Answers: 1

Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1

Chemistry, 22.06.2019 13:30
Apush or pull that moves or changes and object when to objects touch
Answers: 2

Chemistry, 22.06.2019 22:20
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2
You know the right answer?
1.34 milligrams is the same as and...
Questions

Business, 24.07.2019 01:30

History, 24.07.2019 01:30

Social Studies, 24.07.2019 01:30

History, 24.07.2019 01:30

History, 24.07.2019 01:30

Social Studies, 24.07.2019 01:30



Mathematics, 24.07.2019 01:30


Health, 24.07.2019 01:30


Social Studies, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30


Health, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30

Health, 24.07.2019 01:30

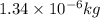 and
and