
Chemistry, 18.12.2020 01:00 aprilkenedy12
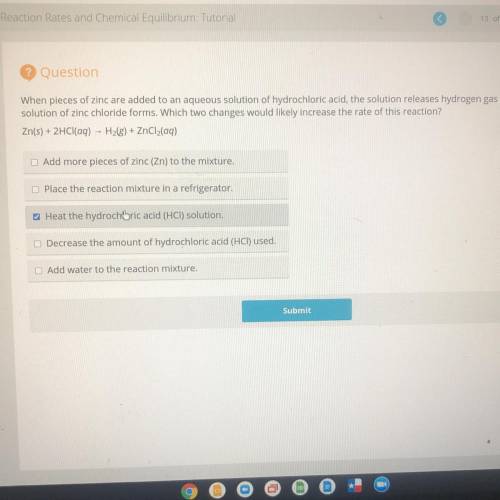
Question
When pieces of zinc are added to an aqueous solution of hydrochloric acid, the solution releases hydrogen gas and a
solution of zinc chloride forms. Which two changes would likely increase the rate of this reaction?
Zn(s) + 2HCl(aq) - Hale) * ZnCl(aq)
Add more pieces of zinc (2n) to the mixture
Place the reaction mixture in a refrigerator,
w Heat the hydrochloric acid (HC) solution.
Decrease the amount of hydrochloric acid (HCI) used,
Add water to the reaction mixture.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:10
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1


Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3
You know the right answer?
Question
When pieces of zinc are added to an aqueous solution of hydrochloric acid, the solution re...
Questions



Mathematics, 25.01.2021 20:20

Mathematics, 25.01.2021 20:20


Mathematics, 25.01.2021 20:20


Mathematics, 25.01.2021 20:20









History, 25.01.2021 20:20



Mathematics, 25.01.2021 20:20



