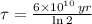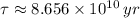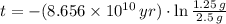Chemistry, 18.12.2020 14:00 kaitlyn2030
14. A sample that originally contained 2.5 g of rubidium-87 now contains 1.25 g. The half-life of rubidium-87 is 6 x 10^10 years. How old is the sample? Is this possible? Why or why not?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 19:40
What is the wavelength of a 3*10^12 hz infrared wave a 3*10^20m b 1* 10^4m c 3*10^-3m d 1*10^-4 m
Answers: 1

Chemistry, 22.06.2019 22:30
Which statement best summarizes the importance of ernest rutherford’s gold foil experiment? it proved that all of john dalton’s postulates were true. it verified j. j. thomson’s work on the atomic structure. it showed that an electron circles a nucleus in a fixed-energy orbit. it showed that a nucleus occupies a small part of the whole atom.
Answers: 1

Chemistry, 23.06.2019 01:30
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
You know the right answer?
14. A sample that originally contained 2.5 g of rubidium-87 now contains 1.25 g. The half-life of ru...
Questions


Biology, 18.03.2021 01:30




Mathematics, 18.03.2021 01:30


Mathematics, 18.03.2021 01:30



Mathematics, 18.03.2021 01:30


English, 18.03.2021 01:30

English, 18.03.2021 01:30


Mathematics, 18.03.2021 01:30

Mathematics, 18.03.2021 01:30

Mathematics, 18.03.2021 01:30

Mathematics, 18.03.2021 01:30

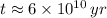 . It is possible, since the definition of half-life is the time taken by the isotope to halve its mass.
. It is possible, since the definition of half-life is the time taken by the isotope to halve its mass. 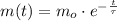 (1)
(1)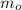 - Initial mass of the isotope, measured in grams.
- Initial mass of the isotope, measured in grams.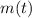 - Current mass of the isotope, measured in grams.
- Current mass of the isotope, measured in grams. 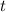 - Time, measured in years.
- Time, measured in years. - Time constant, measured in years.
- Time constant, measured in years.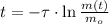
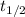 ), measured in years:
), measured in years: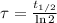 (2)
(2)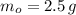 ,
, 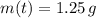 and
and 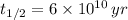 , then the age of the isotope is:
, then the age of the isotope is: