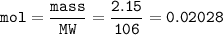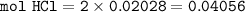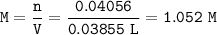Chemistry, 21.12.2020 16:00 ameliaxbowen7
If 38.55 mL of HCl is required to titrate 2.150 g of Na2CO3 according to the following equation, what is the molarity of the HCl solution? Na2CO3 + 2HCl (aq)→ 2NaCl (aq) + CO2 (g) + H20 (1)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:20
In a reaction equation, where are the products located? a.) above the arrow b.) to the right of the arrow c.) to the left of the arrow d.) below the arrow
Answers: 2

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 13:30
Astudent is trying to create a table that compares hypotheses, theories, and laws. hypothesis theory law do scientific researchers formulate it? yes yes yes does it explain why things happen? yes yes no yes yes yes is it used to make predictions? no yes yes which of the following questions would most likely fill the blank in the table? is it an intelligent guess? is it newly formulated? is it based on observations? has it been proved?
Answers: 1
You know the right answer?
If 38.55 mL of HCl is required to titrate 2.150 g of Na2CO3 according to the following equation, wha...
Questions

Advanced Placement (AP), 01.09.2019 06:00

Social Studies, 01.09.2019 06:00


English, 01.09.2019 06:00

Biology, 01.09.2019 06:00




Biology, 01.09.2019 06:00


History, 01.09.2019 06:00





History, 01.09.2019 06:10

Mathematics, 01.09.2019 06:10


Biology, 01.09.2019 06:10