
Calcium Carbonate reacts with dilute hydrochloric acid. The equation for the reaction is shown. CaCo3 + 2Hcl = Cacl2 + H2O + Co2 1 g of calcium Carbonate is added to 50cm3 of 0.05mol/dm3 hydrochloric acid. Which volume of carbon dioxide is made in this reaction?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 10:30
Geothermal energy for industrial use is available almost anywhere. a.true b.false
Answers: 2

Chemistry, 22.06.2019 21:30
Describe at least two advantages and two disadvantages of using hydropower as a source of energy.
Answers: 2

Chemistry, 22.06.2019 23:30
Substance a is a nonpolar liquid and has only dispersion forces among its constituent particles. substance b is also a nonpolar liquid and has about the same magnitude of dispersion forces among its constituent particles. when substance a and b are combined, they spontaneously mix.
Answers: 1
You know the right answer?
Calcium Carbonate reacts with dilute hydrochloric acid. The equation for the reaction is shown. CaCo...
Questions

Mathematics, 08.11.2020 21:40

Biology, 08.11.2020 21:40



Mathematics, 08.11.2020 21:40

English, 08.11.2020 21:40

Advanced Placement (AP), 08.11.2020 21:40

English, 08.11.2020 21:40

Mathematics, 08.11.2020 21:40


History, 08.11.2020 21:40


Biology, 08.11.2020 21:40


Mathematics, 08.11.2020 21:40


English, 08.11.2020 21:50


History, 08.11.2020 21:50

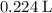 , assuming that this reaction took place under standard temperature and pressure, and that
, assuming that this reaction took place under standard temperature and pressure, and that 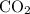 behaves like an ideal gas. Also assume that the reaction went to completion.
behaves like an ideal gas. Also assume that the reaction went to completion.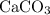 is the limiting reactant. How many moles of
is the limiting reactant. How many moles of 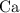 ,
,  , and
, and  on a modern periodic table:
on a modern periodic table: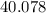 .
.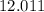 .
.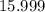 .
.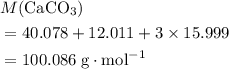 .
.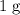 of
of 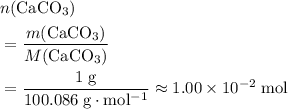 .
.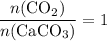 .
.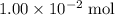 of
of 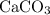 formula would be consumed. That would produce approximately
formula would be consumed. That would produce approximately 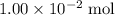 of
of 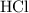 is the limiting reactant.
is the limiting reactant.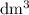 (so as to match the unit of concentration.)
(so as to match the unit of concentration.) .
.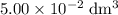 of this
of this 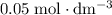
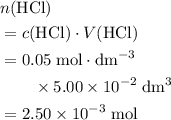 .
.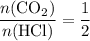 .
.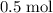 of
of 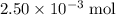 of
of 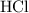 molecules would produce only one-half as many (that is,
molecules would produce only one-half as many (that is, 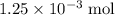 ) of
) of 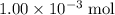 of
of 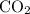 molecules would be produced.
molecules would be produced.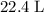 .
.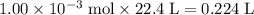 .
.


