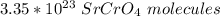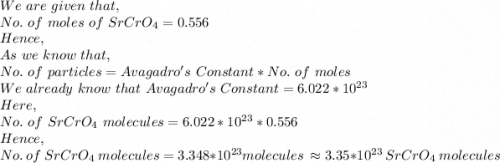Chemistry, 30.12.2020 01:00 montoyaricardo3550
How many molecules of SrCrO4 are in a sample of SrCrO4 0.556 moles?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 14:10
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2

Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

Chemistry, 22.06.2019 21:00
Use the measurements in the table to determine which unidentified metal has the highest density. metal volume mass a 10.5 cm3 122 g b 14.2 cm3 132 g c 16.1 cm3 115 g d 12.7 cm3 126 g
Answers: 2
You know the right answer?
How many molecules of SrCrO4 are in a sample of SrCrO4 0.556 moles?...
Questions



Biology, 14.10.2019 11:50







Mathematics, 14.10.2019 11:50




Social Studies, 14.10.2019 11:50




Physics, 14.10.2019 11:50

Mathematics, 14.10.2019 11:50