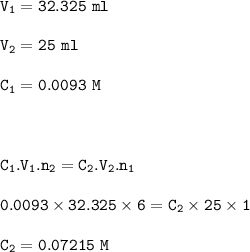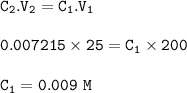Chemistry, 30.12.2020 21:50 lilytimpsonx
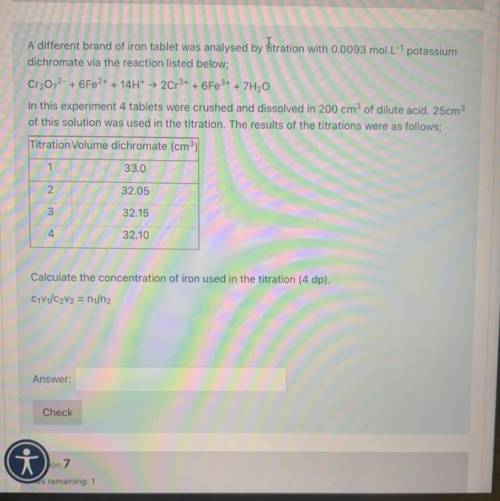
A different brand of iron tablet was analysed by Hitration with 0.0093 mol. L" potassium
dichromate via the reaction listed below;
Cr2O72- + 6Fe2+ + 14H+ + 2Cr3+ + 6Fe3+ + 7H20
In this experiment 4 tablets were crushed and dissolved in 200 cm3 of dilute acid. 25cm3
of this solution was used in the titration. The results of the titrations were as follows;
Titration Volume dichromate (cm)
1
33.0
2
32.05
3
32.15
4
32.10
Calculate the concentration of iron used in the titration (4 dp).
C1V1/C2V2 = n/n2

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2


Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2
You know the right answer?
A different brand of iron tablet was analysed by Hitration with 0.0093 mol. L" potassium
dichromate...
Questions

Mathematics, 04.07.2021 16:20

Mathematics, 04.07.2021 16:20

Social Studies, 04.07.2021 16:20

Mathematics, 04.07.2021 16:20


Mathematics, 04.07.2021 16:20


English, 04.07.2021 16:50

Mathematics, 04.07.2021 16:50

Mathematics, 04.07.2021 16:50

Mathematics, 04.07.2021 16:50

Mathematics, 04.07.2021 16:50






Advanced Placement (AP), 04.07.2021 16:50


Social Studies, 04.07.2021 16:50