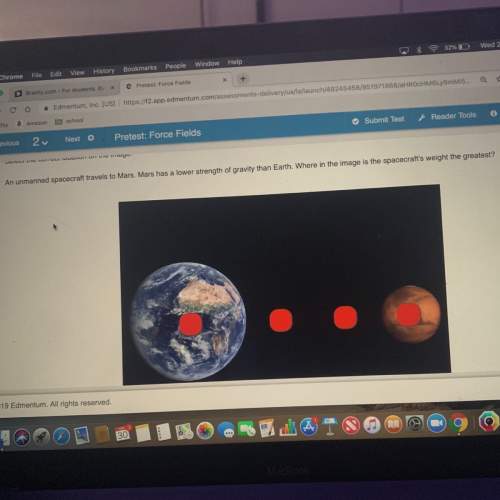
Chemistry, 31.12.2020 19:00 shadow29916
Two solutions initially at 24.60°C are mixed in a coffee cup calorimeter. when a 100.0 ml volume of 0.100 m AgNO3 solution is mixed with a 100.0 ml sample of 0.200 m nacl solution, the temperature in the calorimeter rises to 25.30°C. Determine the dh for the reaction as written below. Assume that the density and heat capacity of the solutions is the same as that of water.
NaCl(aq)+AgNO3(aq) → AgCl(s)+NaNO3(aq)
a. -69 kJ
b. -35 kJ
c. -16 kJ
d. -250 kJ

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The blackbody curve for a star name zeta is shown below. what is the peak wavelength for this star ?
Answers: 1

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2


Chemistry, 23.06.2019 03:00
Analyze the reaction to determine whether the reaction is exothermic or endothermic. explain your reasoning.
Answers: 1
You know the right answer?
Two solutions initially at 24.60°C are mixed in a coffee cup calorimeter. when a 100.0 ml volume of...
Questions

Chemistry, 24.05.2021 07:20


Mathematics, 24.05.2021 07:20


Mathematics, 24.05.2021 07:20


Mathematics, 24.05.2021 07:20


Social Studies, 24.05.2021 07:20


Mathematics, 24.05.2021 07:20



Mathematics, 24.05.2021 07:20

Mathematics, 24.05.2021 07:20


Mathematics, 24.05.2021 07:20


Chemistry, 24.05.2021 07:20

Health, 24.05.2021 07:20