
Chemistry, 03.01.2021 21:00 averystricker7837
PLEASE HELP ME I NEED TO FINISH BY 11 PM
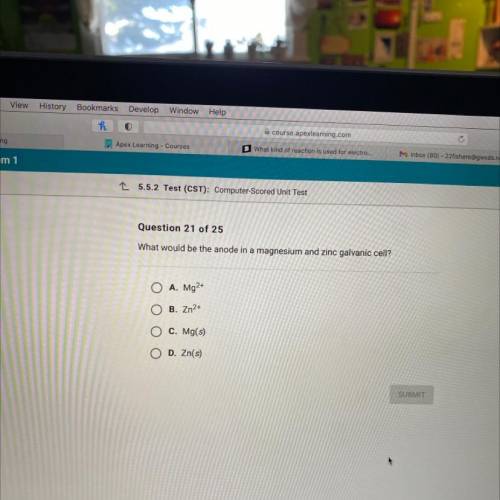
What would be the anode in a magnesium and zinc galvanic cell?
A. Mg2+
B. Zn2+
C. Mg(s)
D. Zn(s)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:20
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2

Chemistry, 22.06.2019 01:00
Which of the following is always a reactant in a combustion reaction? oxygen nitrogen hydrogen carbon
Answers: 1

Chemistry, 22.06.2019 06:00
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
You know the right answer?
PLEASE HELP ME I NEED TO FINISH BY 11 PM
What would be the anode in a magnesium and zinc galvanic c...
Questions


Physics, 29.12.2019 06:31

English, 29.12.2019 06:31


Mathematics, 29.12.2019 06:31

Law, 29.12.2019 06:31

Social Studies, 29.12.2019 06:31

Mathematics, 29.12.2019 06:31


Mathematics, 29.12.2019 06:31



Biology, 29.12.2019 06:31


Computers and Technology, 29.12.2019 06:31

Mathematics, 29.12.2019 06:31




Mathematics, 29.12.2019 06:31



