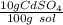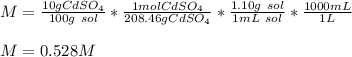Chemistry, 07.01.2021 18:30 arieltaylor3924
What is the molarity of a solution of 10% by mass cadmium sulfate, CdSO4 (molar mass = 208.46 g/mol) by mass? The density of the solution is 1.10 g/mL.
a. 0.528 M
b. 0.436 M
c. 0.479 M
d. 0.048 M
e. 22.9 M

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1

Chemistry, 22.06.2019 19:30
Describe the forces both attractive and repulsive that occur as two atoms move closer together.
Answers: 1

Chemistry, 23.06.2019 03:00
What volume does 1.70 ×10–3 mol of chlorine gas occupy if its temperature is 20.2 °c and its pressure is 795 mm hg?
Answers: 3

Chemistry, 23.06.2019 11:50
It takes 155. kj/mol to break a fluorine-fluorine single bond. calculate the maximum wavelength of light for which a flouine-flouring single bond could be broken by absorbing a single photon
Answers: 1
You know the right answer?
What is the molarity of a solution of 10% by mass cadmium sulfate, CdSO4 (molar mass = 208.46 g/mol)...
Questions

Social Studies, 15.04.2020 20:13


Mathematics, 15.04.2020 20:13


Biology, 15.04.2020 20:13

Physics, 15.04.2020 20:13

Mathematics, 15.04.2020 20:13








Mathematics, 15.04.2020 20:13





Mathematics, 15.04.2020 20:13