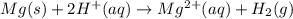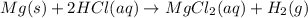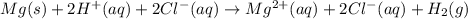Mg(s)+2HCl(aq) → MgCl2(aq)+H2(g)
In an experiment, a student places a small piece of pure Mg(s) into a beaker containing 250.mL of 6.44MHCl(aq) . A reaction occurs, as represented by the equation above.
(a) Write the balanced net ionic equation for the reaction between Mg(s) and HCl(aq) .

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Write a paragraph that provides examples of each stage of volcanic activity, a description of the volcano, and facts about each stage.
Answers: 1


Chemistry, 22.06.2019 10:00
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1

You know the right answer?
Mg(s)+2HCl(aq) → MgCl2(aq)+H2(g)
In an experiment, a student places a small piece of pure Mg(s)...
Questions


Mathematics, 25.07.2020 02:01




Mathematics, 25.07.2020 02:01


Mathematics, 25.07.2020 02:01

Mathematics, 25.07.2020 02:01









English, 25.07.2020 02:01


Mathematics, 25.07.2020 02:01