
Chemistry, 08.01.2021 18:20 epicsushiman1
A flask containing 855 grams of water was heated. If the temperature of the water was raised from 21.0 to 85.0 degrees Celsius, how much heat (in joules) did the water absorb? the specific heat capacity of liquid water is 4.184J/g per C

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 23.06.2019 10:50
Which compound should undergo substitution of the bromine by phenolate anion? draw the structure of the organic product?
Answers: 1

Chemistry, 23.06.2019 16:00
Water is called the universal solvent because more substances dissolve in water than in any other chemical. this has to do with the polarity of each water molecule. the hydrogen side of each water (h2o) molecule carries a slight positive electric charge, while the oxygen side carries a slight negative electric charge.
Answers: 3

Chemistry, 23.06.2019 16:00
Challenge question: this question is worth 6 points. as you saw in problem 9 we can have species bound to a central metal ion. these species are called ligands. in the past we have assumed all the d orbitals in some species are degenerate; however, they often are not. sometimes the ligands bound to a central metal cation can split the d orbitals. that is, some of the d orbitals will be at a lower energy state than others. ligands that have the ability to cause this splitting are called strong field ligands, cnâ’ is an example of these. if this splitting in the d orbitals is great enough electrons will fill low lying orbitals, pairing with other electrons in a given orbital, before filling higher energy orbitals. in question 7 we had fe2+, furthermore we found that there were a certain number (non-zero) of unpaired electrons. consider now fe(cn)6 4â’: here we also have fe2+, but in this case all the electrons are paired, yielding a diamagnetic species. how can you explain this?
Answers: 2
You know the right answer?
A flask containing 855 grams of water was heated. If the temperature of the water was raised from 21...
Questions

Mathematics, 10.03.2020 09:16




Mathematics, 10.03.2020 09:16



Mathematics, 10.03.2020 09:16

Mathematics, 10.03.2020 09:16






Computers and Technology, 10.03.2020 09:16



Mathematics, 10.03.2020 09:16


English, 10.03.2020 09:16

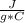 m=855 gΔT= Tfinal - Tinitial= 85 °C - 21°C= 64 °C
m=855 gΔT= Tfinal - Tinitial= 85 °C - 21°C= 64 °C

