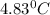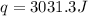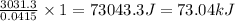2
3 of 6 - SCH4U
Date: Name(s):
4. HCl is a corrosive colourless gas that dissolves rea...

Chemistry, 09.01.2021 06:20 saleenhernandez83
2
3 of 6 - SCH4U
Date: Name(s):
4. HCl is a corrosive colourless gas that dissolves readily in water.
Aqueous HCl reacts with NaOH to form water and NaCl. In a simple
calorimeter, a 100.00 mL sample of 0.415 mol/L HCl(aq) is mixed
with 50.00 mL of excess NaOH(aq). During the reaction, there is a
rise in temperature by 4.83 °C. Calculate the molar enthalpy change
(in kJ/mol) for the above reaction. SHOW ALL YOUR WORK.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
This chart represents the melting point of several substance. what besy explains the high melting point of the salt?
Answers: 2

Chemistry, 22.06.2019 09:30
Right anwser gets marked brainliest newton's discovery concerning how fast an object will change speed is the: 1st law 2nd law 3rd law universal gravitation
Answers: 1

Chemistry, 22.06.2019 10:30
How do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 2

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical systems? a) water dissolves nonpolar ionic compounds. b) water dissociates ionic compounds. c) water dissociates covalent molecules. d) water dissolves nonpolar covalent substances.
Answers: 1
You know the right answer?
Questions




Biology, 23.03.2021 16:20




Mathematics, 23.03.2021 16:20






History, 23.03.2021 16:20


Mathematics, 23.03.2021 16:20



French, 23.03.2021 16:20


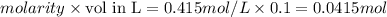
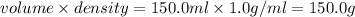
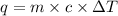
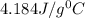
 = change in temperature =
= change in temperature =