
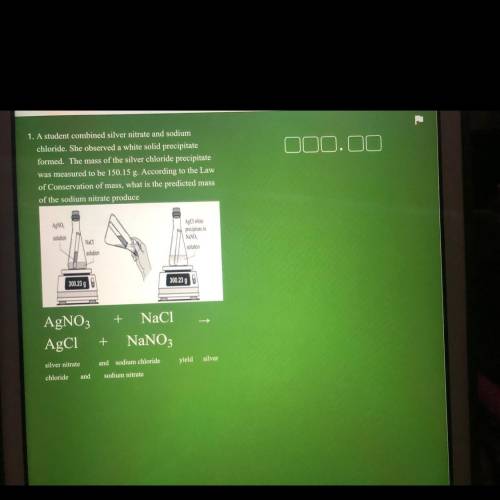
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
formed. The mass of the silver chloride precipitate
was measured to be 150.15 g. According to the Law
of Conservation of mass, what is the predicted mass
of the sodium nitrate produce
AgNO
AgCl white
precipitate in
solution
Naci
NaNO
solution
solution
300.23 g
300.23 90
+ NaCl
AgNO3
AgC1
+
NaNO3
silver nitrate
and sodium chloride
yield silver
chloride
and
sodium nitrate

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:50
If the mass of the products measured 120g what would the mass of the reactants a. 30g b. 60g c. 120g d. 240g
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1

Chemistry, 23.06.2019 00:00
How many atoms or molecules are there in a mole of a substance?
Answers: 1
You know the right answer?
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
Questions

Mathematics, 23.07.2020 14:01

English, 23.07.2020 14:01





Mathematics, 23.07.2020 14:01



Physics, 23.07.2020 14:01

Engineering, 23.07.2020 14:01

History, 23.07.2020 14:01

Chemistry, 23.07.2020 14:01



Mathematics, 23.07.2020 14:01






