
Nitrogen dioxide can be made with the reaction of nitrogen monoxide and oxygen.
Assuming that the reaction is at equilibrium, what effect would a decrease in the concentration of NO_2 have on the equilibrium position once the equilibrium is reestablished?
A. An increase in the amount of begin mathsize 14px style NO subscript 2 end style.
B. An increase in the amount of begin mathsize 14px style straight O subscript 2 end style.
C. No change in the amount of begin mathsize 14px style straight O subscript 2 end style.
D. An increase in the amount of NO.
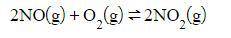

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Mitosis is a type of cell division that produces cells that are identical to the parent cell. meiosis is a different type of cell division that produces cells that carry have a genetic material of the parent cell. based on the information provided how do the purpose of mitosis and meiosis differ
Answers: 3

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1
You know the right answer?
Nitrogen dioxide can be made with the reaction of nitrogen monoxide and oxygen.
Assuming that the r...
Questions






History, 16.10.2019 19:50



Mathematics, 16.10.2019 19:50

Social Studies, 16.10.2019 19:50


Mathematics, 16.10.2019 19:50


Mathematics, 16.10.2019 19:50


Social Studies, 16.10.2019 19:50


Health, 16.10.2019 19:50

History, 16.10.2019 19:50




