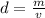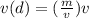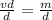Chemistry, 10.01.2021 22:40 makennskyee7114
Density is defined as mass per unit volume, or D = m/v. Solve the density
equation for the variable v: *
v = D + m
v = D/m
v = D * m
v= m/D

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Asolution is made by dissolving 25.5 grams of glucose (c6h12o6) in 398 grams of water. what is the freezing point depression of the solvent if the freezing point constant is -1.86 °c/m? show all of the work needed to solve this problem.
Answers: 1

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1
You know the right answer?
Density is defined as mass per unit volume, or D = m/v. Solve the density
equation for the variable...
Questions

Mathematics, 05.10.2020 15:01


Mathematics, 05.10.2020 15:01



Mathematics, 05.10.2020 15:01

Mathematics, 05.10.2020 15:01




English, 05.10.2020 15:01


Health, 05.10.2020 15:01


Mathematics, 05.10.2020 15:01

English, 05.10.2020 15:01


World Languages, 05.10.2020 15:01