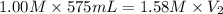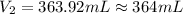Chemistry, 11.01.2021 16:10 makailaaa2
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do this by pouring out some 1.58M aqueous sodium carbonate stock solution into a graduated cylinder and diluting it with distilled water. Calculate the volume in mL of the sodium carbonate stock solution that the chemist should pour out. Round your answer to 3 significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2

Chemistry, 22.06.2019 18:00
The human activities in two locations are described below: location a: rampant use of plastic containers location b: excessive use of pesticides and fertilizers which statement is most likely true? location a will have poor air quality because plastic is biodegradable. location a will experience water scarcity because plastic absorbs moisture. the population of honeybees will increase in location b because production of crops will increase. the population of fish in location b will decrease because the water is contaminated.
Answers: 1
You know the right answer?
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do th...
Questions









Biology, 03.08.2019 10:50



History, 03.08.2019 10:50

Biology, 03.08.2019 10:50



Social Studies, 03.08.2019 10:50


Mathematics, 03.08.2019 10:50


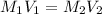
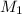 = molarity of aqueous sodium carbonate
= molarity of aqueous sodium carbonate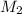 = molarity of aqueous sodium carbonate stock solution
= molarity of aqueous sodium carbonate stock solution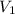 = volume of aqueous sodium carbonate
= volume of aqueous sodium carbonate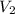 = volume of aqueous sodium carbonate stock solution
= volume of aqueous sodium carbonate stock solution