
Chemistry, 12.01.2021 01:00 danielanderson12
A sample of He - gas has 5.03 x 10 23He atoms. What volume is occupied by this sample at s. t.p?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 05:00
Which position represents spring in the southern hemisphere? a) b) c) d)
Answers: 2

You know the right answer?
A sample of He - gas has 5.03 x 10 23He atoms. What volume is occupied by this sample at s. t.p?...
Questions

Social Studies, 30.10.2019 01:31

Mathematics, 30.10.2019 01:31




History, 30.10.2019 01:31








History, 30.10.2019 01:31


Chemistry, 30.10.2019 01:31



History, 30.10.2019 01:31

Mathematics, 30.10.2019 01:31

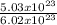 = 0.84mole of He
= 0.84mole of He

