
Chemistry, 12.01.2021 16:50 zafarm2oxgpmx
Calculate the morality for the following
10.5 kg of Na2 SO4. 10H2O in 18.60 L

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
This image is an example of a(n) a) atom. b) compound. c) mixture. d) molecule.
Answers: 1

Chemistry, 22.06.2019 19:00
Avolleyball player hit a ball with a mass of 0.25 kg. the average acceleration of the ball is 15.5 m/s². how much force did the volleyball player apply to the ball? 62.0 n 3.87 n 62.0 m/s² 3.87 m/s²
Answers: 2

Chemistry, 23.06.2019 05:00
Asolution is made by dissolving 2.3 moles of sodium chloride (nacl) in 0.155 kilograms of water. if the molal boiling point constant for water (kb) is 0.51 °c/m, what would be the boiling point of this solution? show all the steps taken to solve this problem.
Answers: 1

Chemistry, 23.06.2019 06:20
Type the correct answer in each box.balance the chemical equation.__ n203 ➡️ __ n2 +__ o2
Answers: 1
You know the right answer?
Calculate the morality for the following
10.5 kg of Na2 SO4. 10H2O in 18.60 L...
10.5 kg of Na2 SO4. 10H2O in 18.60 L...
Questions


Mathematics, 17.12.2020 23:50

Arts, 17.12.2020 23:50

Geography, 17.12.2020 23:50

Social Studies, 17.12.2020 23:50

Mathematics, 17.12.2020 23:50


Biology, 17.12.2020 23:50

Mathematics, 17.12.2020 23:50



Mathematics, 17.12.2020 23:50





Mathematics, 17.12.2020 23:50

Mathematics, 17.12.2020 23:50

Mathematics, 17.12.2020 23:50

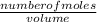
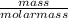
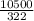 = 32.6mole
= 32.6mole 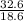 = 1.75M
= 1.75M

