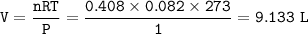Chemistry, 13.01.2021 07:10 tawna6988owtjg6
Trimethylamine, (CH3)2N is a weak base (K6 = 6.3 x 10-5). What volume of this gas, measured at STP, must be dissolved in 2.5 L of solution to give that solution a pOH of 2.50? PLEASE HELP ITS DUE IN 20 mins :

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1

Chemistry, 23.06.2019 07:00
Agas has an empirical formula ch4. 0.16g of the gas occupies a volume of 240cm^3 what is the molecular formula of the me anyone who !
Answers: 1
You know the right answer?
Trimethylamine, (CH3)2N is a weak base (K6 = 6.3 x 10-5). What volume of this gas, measured at STP,...
Questions


Mathematics, 19.03.2021 22:10

Chemistry, 19.03.2021 22:10

Mathematics, 19.03.2021 22:10

Mathematics, 19.03.2021 22:10






Mathematics, 19.03.2021 22:10






History, 19.03.2021 22:10


Mathematics, 19.03.2021 22:10

![\tt [OH^-]=10^{-2.5}=0.0032=3.2\times 10^{-3}](/tpl/images/1031/5783/b6571.png)
![\tt [OH^-]=\sqrt{Kb.M}\\\\(3.2\times 10^{-3})^2=6.3\times 10^{-5}\times M\\\\M=0.163](/tpl/images/1031/5783/8f3e1.png)