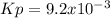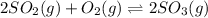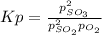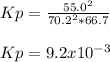Sulfur dioxide and oxygen react to form sulfur trioxide, like this: (g) (g) (g) Also, a chemist finds that at a certain temperature the equilibrium mixture of sulfur dioxide, oxygen, and sulfur trioxide has the following composition: compound pressure at equilibrium Calculate the value of the equilibrium constant for this reaction. Round your answer to significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:30
How many joules of heat are absorbed to raise the temperature of 650 grams of water from 5.00c to it's boiling point, 100c
Answers: 1

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1


You know the right answer?
Sulfur dioxide and oxygen react to form sulfur trioxide, like this: (g) (g) (g) Also, a chemist find...
Questions





History, 20.10.2020 16:01


English, 20.10.2020 16:01





Mathematics, 20.10.2020 16:01


Health, 20.10.2020 16:01

Mathematics, 20.10.2020 16:01