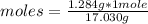Chemistry, 13.01.2021 15:40 anavallesdemiguel2
Calculate the moles of ammonia present in a 1.284 g sample if the molar mass of ammonia is 17.030 g/mol.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1


Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
Calculate the moles of ammonia present in a 1.284 g sample if the molar mass of ammonia is 17.030 g/...
Questions


Biology, 26.09.2019 19:00





Spanish, 26.09.2019 19:00




English, 26.09.2019 19:00


Business, 26.09.2019 19:00





Mathematics, 26.09.2019 19:00