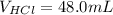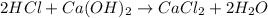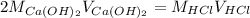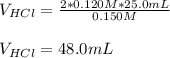Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 22.06.2019 20:40
What effect would average population growth have on land usage? a. urban use of land would rise to more than 30 percent of available land. b. industrial use of land would rise to more than 30 percent of available land. c. the percentage of available land used as cropland would stay the same. d. cropland would fall to about 10 percent of available land.
Answers: 1

Chemistry, 22.06.2019 22:30
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1

Chemistry, 22.06.2019 23:00
The data below were determined for the reaction shown below. s2o82– + 3i – (aq) → 2so42– + i3– expt. # [s2o82–] (m) [i –] (m) initial rate 1 0.038 0.060 1.4 × 10 – 5 m/s 2 0.076 0.060 2.8 × 10 – 5 m/s 3 0.076 0.030 1.4 × 10 – 5 m/s the rate law for this reaction must be:
Answers: 1
You know the right answer?
A sample of 25.0 mL of 0.120 M Ca(OH)2(aq) is titrated with 0.150 M HCl(aq). What volume of HCl(aq)...
Questions



History, 21.12.2020 04:10

Geography, 21.12.2020 04:10

Mathematics, 21.12.2020 04:10




English, 21.12.2020 04:10

History, 21.12.2020 04:10

English, 21.12.2020 04:10

Mathematics, 21.12.2020 04:10



Mathematics, 21.12.2020 04:10

Physics, 21.12.2020 04:10


Mathematics, 21.12.2020 04:10